|
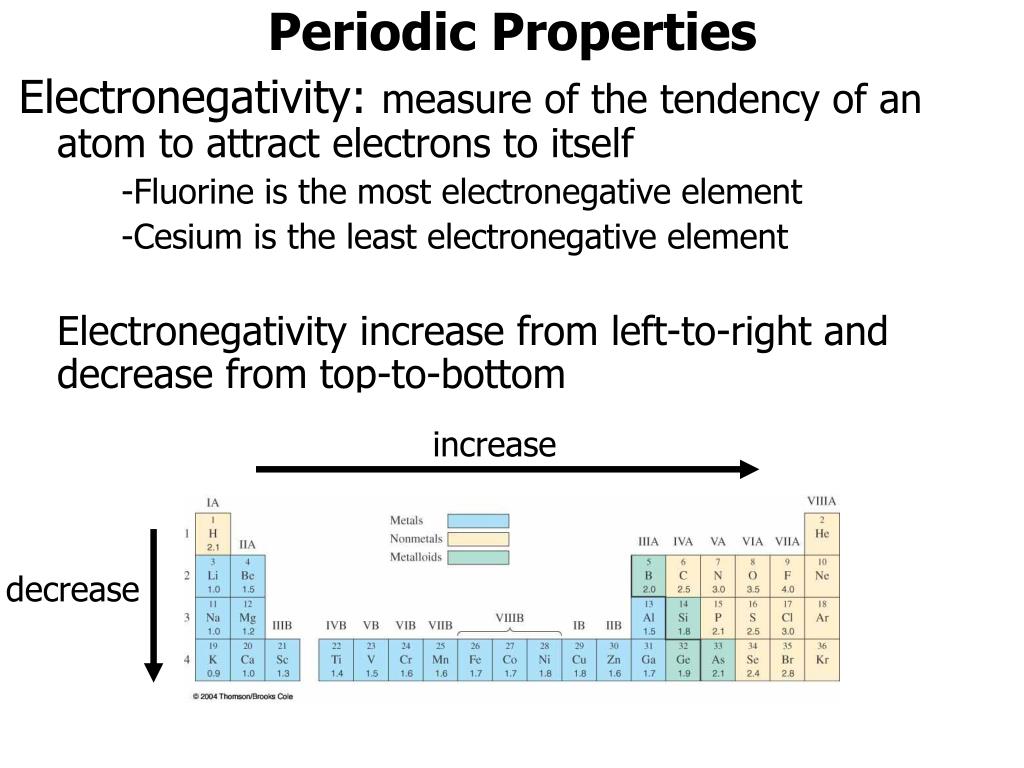
As you can see, electronegativities generally increase from left to right across a period and decrease down a group. A value of 4.0 is assigned to fluorine, the most electronegative element. In contrast to intramolecular forces, such as the covalent bonds that hold atoms together in molecules and polyatomic ions, intermolecular forces hold molecules together in a liquid or solid.Intermolecular forces are generally much weaker than covalent bonds. The periodic table below shows the Pauling electronegativity scale. Than the other atom, the electrons will not be shared and an ionic bond will If one atom is overwhelmingly more electronegative If one atom is more electronegative, the electrons of the bond are Two atoms of the bond are of equal electronegativity, the electrons are equally Team is no longer able to hold onto the rope and the entire rope ends up on If one team is overwhelmingly stronger, the weaker If one team is stronger, the rope is pulled Will not be shared at all the more electronegative atom will "take" them resulting If the difference in electronegativity is large enough, the electrons Of the atoms (because it is more electronegative), the electrons will be unequally Major trends are electronegativity, ionization energy, electron affinity, atomic radius. If the electrons of a bond are more attracted to one Answer (1 of 4): A2A Let’s first define electronegativity: Electronegativity is the measure of tendency of a nucleus of an atom to attract a bonding pair of electrons. Periodic trends are patterns in elements on the periodic table. 
If atoms bonded together have the same electronegativity, the shared electrons In your view, is it important and what.An atom's ability to attract the shared electrons of a covalent bond to itself.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
